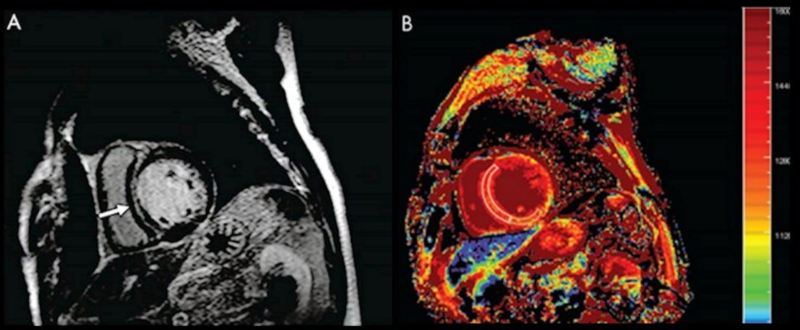
Molecular diagnostics for early detection of vascular events
Cardiovascular diseases remain the leading cause of mortality worldwide. Myocardial infarction and ischemic stroke are characterized by sudden onset and a high probability of disability or death. A central objective of modern cardiology and neurology is the identification of pathological processes at the earliest possible stage, when tissue damage may still be limited or partially reversible. In this context, biomarkers play a critical role in early diagnosis and risk assessment.
Biomarkers are measurable biological molecules whose concentration changes in response to ischemia, inflammation, thrombosis, or cellular injury. Their detection in blood or other biological fluids provides objective information about ongoing pathological processes. In the setting of myocardial infarction, cardiac-specific troponins are considered the most clinically relevant biomarkers.
High-sensitivity troponin I and troponin T assays have transformed the diagnosis of acute coronary syndromes. These structural proteins are released into the circulation following damage to cardiomyocytes. Modern analytical techniques allow detection of minimal elevations within one to three hours after the onset of ischemia. This rapid identification significantly reduces the time to diagnosis and initiation of reperfusion therapy.
In addition to troponins, other markers have been evaluated in the assessment of myocardial injury. Creatine kinase-MB was previously widely used but has largely been replaced by more specific indicators. Copeptin, a surrogate marker of endogenous stress response, has been investigated as an adjunct in the very early phase of acute coronary events, particularly when troponin levels are still within reference limits.
The identification of early biomarkers in ischemic stroke presents additional challenges. Although the brain is highly sensitive to hypoxia, many neuronal proteins are not readily detectable in peripheral circulation. Molecules such as neuron-specific enolase, S100B protein, and glial fibrillary acidic protein have been studied as potential indicators of neuronal and glial injury. Elevation of these markers may reflect the extent of cerebral tissue damage.
Inflammatory and thrombotic markers also contribute to risk stratification. Elevated C-reactive protein, interleukins, and fibrinogen levels indicate systemic inflammation and may signal instability of atherosclerotic plaques. D-dimer reflects activation of the coagulation cascade and is associated with an increased risk of thrombotic complications.
An emerging field involves the study of circulating microRNAs. These small non-coding RNA molecules regulate gene expression and exhibit disease-specific expression patterns. Certain microRNAs demonstrate relative specificity for cardiac or neural tissue injury, suggesting potential use as early diagnostic tools. However, their clinical implementation requires further validation and methodological standardization.
Biomarkers are valuable not only for diagnosing acute events but also for predicting future risk. Natriuretic peptides reflect myocardial stress and may indicate an elevated likelihood of heart failure and cardiovascular events. Lipoprotein(a) has been identified as an independent risk factor for atherothrombosis. Comprehensive evaluation of such markers allows the development of individualized cardiovascular risk profiles.
The integration of laboratory biomarkers with imaging modalities enhances diagnostic precision. In suspected myocardial infarction, electrocardiographic and echocardiographic findings are interpreted alongside biochemical results. In stroke, computed tomography and magnetic resonance imaging remain essential for confirmation, while biomarkers may provide complementary information regarding pathophysiology and prognosis.
Multiparametric testing panels combining several biomarkers represent a promising strategy. By simultaneously assessing ischemia, inflammation, thrombosis, and cellular necrosis, such panels may improve diagnostic sensitivity and specificity compared to single-marker approaches.
Serial measurement is another important principle. Repeated assessment over defined intervals helps differentiate acute dynamic changes from chronically elevated baseline levels associated with comorbidities such as chronic kidney disease or heart failure. This approach is particularly relevant when using high-sensitivity troponin assays.
In conclusion, the development of high-sensitivity and molecularly targeted biomarkers has significantly advanced the early detection of myocardial infarction and stroke. Prompt identification of ischemic injury facilitates timely intervention, reduces the extent of tissue damage, and improves clinical outcomes. Ongoing research aims to refine diagnostic accuracy and expand preventive strategies through earlier and more precise molecular assessment.
Write a review
Required fields are marked with *
Categories
- News (42)
- Therapy (31)
- GP (23)
- Endocrinology (8)
- Cardiology (8)
- Ortopedics (4)
- Dermatology (3)
- urology (1)
- Check-up (1)
- Ultrasound (1)
Articles
Archive
- February 2026 (8)
- January 2026 (8)
- December 2025 (6)
- November 2025 (6)
- October 2025 (6)
- September 2025 (6)
- August 2025 (7)
- July 2025 (4)
- June 2025 (11)
- May 2025 (9)
Categories
- News (42)
- Therapy (31)
- GP (23)
- Endocrinology (8)
- Cardiology (8)
- Ortopedics (4)
- Dermatology (3)
- urology (1)
- Check-up (1)
- Ultrasound (1)








Comments (0)